Chapter 13: Chemical Bonding
Chater 13 was basically about how electrons in atoms form new substances using chemical bonding. Chemical bonding is the combining of atoms to form molecules, so basiclly what that means is that in order to form new bonds in atoms and substances you would need to have new chemical bonds.
VOCAB
1. CHEMICAL BONDING: the combinig of atoms to form molecules or ionic compounds
2. VALENCE ELECTRON: An electron that is found in the outermost shell of an atom and that determines the atoms chemical propereties
3. IONIC BONDING: A bond that forms when electrons are transferred from one atom to another atom
4. IONS: Are charged particles that form when atoms gain or lose electrons
5. CRYSTAL LATTICE: The regular pattern in which a crystal is arranged
6. COVALENT BONDING: A bond formed when atoms share one or more pairs of electrons
7. MOLECULE: Consist of two or more atoms joined in a definite ratio.
8. METALLIC BOND: A bond formed by the attraction between positively charged metal ions and the electrons in the metal
2. VALENCE ELECTRON: An electron that is found in the outermost shell of an atom and that determines the atoms chemical propereties
3. IONIC BONDING: A bond that forms when electrons are transferred from one atom to another atom
4. IONS: Are charged particles that form when atoms gain or lose electrons
5. CRYSTAL LATTICE: The regular pattern in which a crystal is arranged
6. COVALENT BONDING: A bond formed when atoms share one or more pairs of electrons
7. MOLECULE: Consist of two or more atoms joined in a definite ratio.
8. METALLIC BOND: A bond formed by the attraction between positively charged metal ions and the electrons in the metal
CHAPTER 13 LESSON 1:ELECTRONS AND CHEMICAL BONDING
This lesson was basiclly about how electrons can tell you how valence electrons and the atomic number in a atom tell how they bond and what do they bond to. The atomic number in a atom are the protons in a atom, but since atoms do not have a charge the atomic number also tells you the number of electrons in a atom. Some key points of section 1 includes that to have a full outermost energy level of an atom you would need to have 8 valence electrrons, but not all atoms need 8 valence electrons to have a full outermost energy level including helium. This is paragragh sums up the things and key facts you need to know for section 1.
CHAPTER 13 LESSION 2:IONIC BONDS
Lesson 2 basiclly tells you about ionic bonds and how they form. Ionic bonds are bonds that form when electrons are transferred from one atom to another(referr back to vocabulary). Ionic bonds form during chemical changes when atoms pull electrons away from other atoms. The atoms that lose electrons form ions (which are charged particles that form when atoms gain or lose electrons) that have fewer electrons than protons. Atoms of most metals have valence electrons, but metal atoms lose these valence electrons and form positive ions. Only a small amount of energy is needed to take electrons from atoms in groups one and two. An ionic bond will form between a metal and nonmetal ion only if the nonmetal releases more energy that is needed to take electrons from metal.
CHAPTER 13 LESSON 3:COVALENT AND METALLIC BONDS
`Covalent bonds form when atoms share pairs of electrons. substances containing covalent bonds consist of particales called molecules. A molecule is the smallest unit of any substance that keeps all properties of that substance. One way to represent atoms atoms and molecules is to use a electron dot diagram. Electron dot diagrams show only the valence electrons of an atom, but they can help you predict how atoms bond with one another. Electron dot diagrams show the symbol of the element, and how many valence electrons it has. Molecules that are made up of two atoms are called diatomic molecules. Elements that are made up of diatomic molecules are called diatomic elements. Examples of diatomic elements include: hydrogen, oxygen, nitrogen, halogens, flourine, chlorine, bromine, and iodine are diatomic elements. Metals can be reshaped only because of their metallic bonds. Metallic bonds are bonds that form by bonding of positively charged metal ions and electrons in the metal. Positivley charged metal ions form when metal atoms lose electrons. Metallic bonding also gives metals their properties like: conducting electrical current, or ductility and malleability. When a a piece of metal is bent, some of the metal ions are forced closer together, so since electrons constantly move around and between metal ions. The electrons keep their bonds no matter what the metal reshapes into. This is how metal objects can be bent without being broken.
COVALENT BONDING
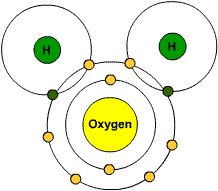
In the picture to left the two hydrogen atoms are sharing valence electrons from the oxygen atoms, so that they can have a full outer shell.
ionic bonds

The picture to the left shows a cartoon about a cat imitating james bond, and relating to how ionic bonds take electrons.
metallic bond
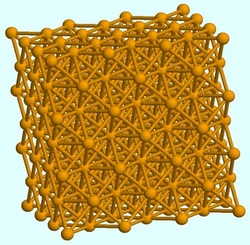
In the picture shows a metallic bond structure,that can reshape without breaking. click here to find out more
